HOME / Departments / Chemistry / Physical Coordination Chemistry
Physical Coordination Chemistry
-
- OHBA Masaaki, Professor
- OHTANI Ryo, Associate Professor
- Benjamin Le Ouay, Assistant Professor
- In the field of molecular materials, one of the important targets is the development of multi-functional material combining different properties and functions in a synergic way. Our group focuses on “coordination polymers” and “liposome” as a meso-scale platform for interlocking various functions and properties. The coordination polymers can provide functional space that consists of flexible, highly ordered and designable frameworks based on coordination bonds. The frameworks can be incorporated magnetic, electrical, optical and other properties, and also can adsorb molecules in the void of structure. The liposome, a spherical vesicle composed of a phospholipid bilayer, provides hierarchical composite with selectively incorporating different functional molecules into the hydrophilic inner water phase, hydrophobic lipid bilayer, and inner and outer surfaces. We are exploring advanced functions using such spaces with synergically linking plural different properties of components.
Research Contents
The research in the Physical Coordination Chemistry Laboratory aims at the synthesis of novel integrated compounds and the development of their functions and properties based on "molecular assembly systems", in which coordination compounds (metal complexes) are used as key compounds. One of the challenges in molecular science is to control the spatial arrangement of individual molecules to achieve higher order organization and to make them function dynamically and cooperatively. Such higher-order organization of molecules plays an important role in electron transfer systems such as photosynthesis and biomolecular motors such as ATPase in biological systems. Metal complexes, on the other hand, are a group of compounds that lie between inorganic and organic compounds. The integrated system of metal complexes, which combines the "excellent individual performance and elemental diversity of inorganic materials" and the "excellent molecularity and designability of organic materials" at the molecular level, makes it possible to construct novel steric and electronic structures and express unique functions and properties that cannot be realized with conventional organic or inorganic materials alone. By skillfully controlling molecular structures and molecular assemblies, we construct unique interactive spaces and conduct research aimed at discovering new phenomena and creating new functions and properties by utilizing various external fields (magnetic fields, electric fields, light, chemical substances, etc.).
The following two major research themes are the main pillars of our research.
- Construction of advanced functional space through higher-order organization of metal complex molecules themselves
- Development of functional materials by compositing metal complex molecules
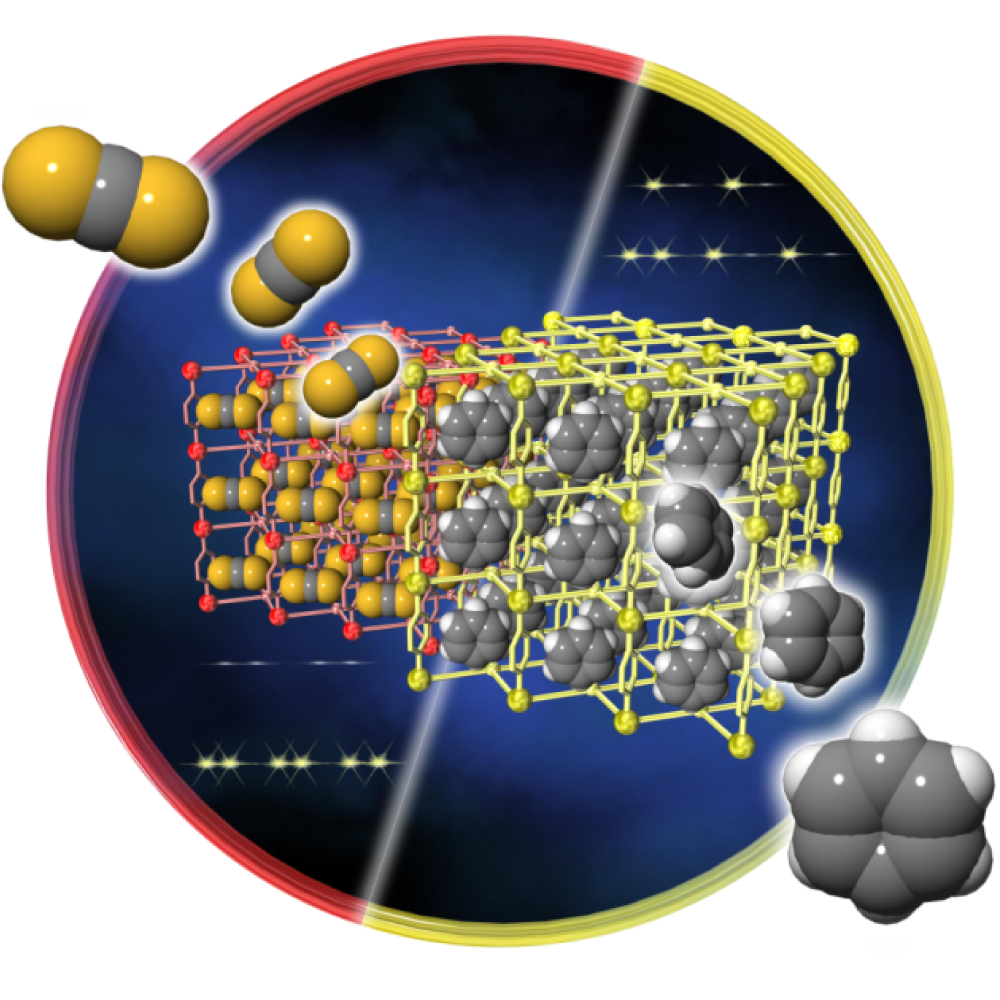
In Theme I, we focus on "Porous Coordination Polymer (PCP)" and "Metal-Organic Framework (MOF)" with nano-sized flexible pores. By incorporating magnetism, conductivity, polarity or luminescence properties into the framework structure, we aim to develop novel physical properties related to molecular adsorption and the behavior of adsorbed molecules in the pore. For example, we have studied the magnetic property conversion by selective guest adsorption/desorption (Chem. Commun., 57, 5211 (2021) etc.), the selective molecular adsorption and emission property conversion using flexible pore structures (Inorg. Chem., 60, 6140 (2021) etc.), and the chemical sensors that respond to specific substances (Chem. Commun., 56, 12961 (2020) etc.). We are also investigating the construction of polar structures and the development of specific properties through the accumulation of metal complexes (J. Am. Chem. Soc., 146, 1476 (2024)), the control of the phase transition behavior of metal complex assemblies and the elucidation of its mechanism (Angew. Chem. Int. Ed., 62, e202306853 (2023), etc.), the negative thermal expansion of the crystal lattice of MOFs (Inorg. Chem., 61, 21123 (2022), etc.) and the development of a molecular spin-qubit based on a metal complex.
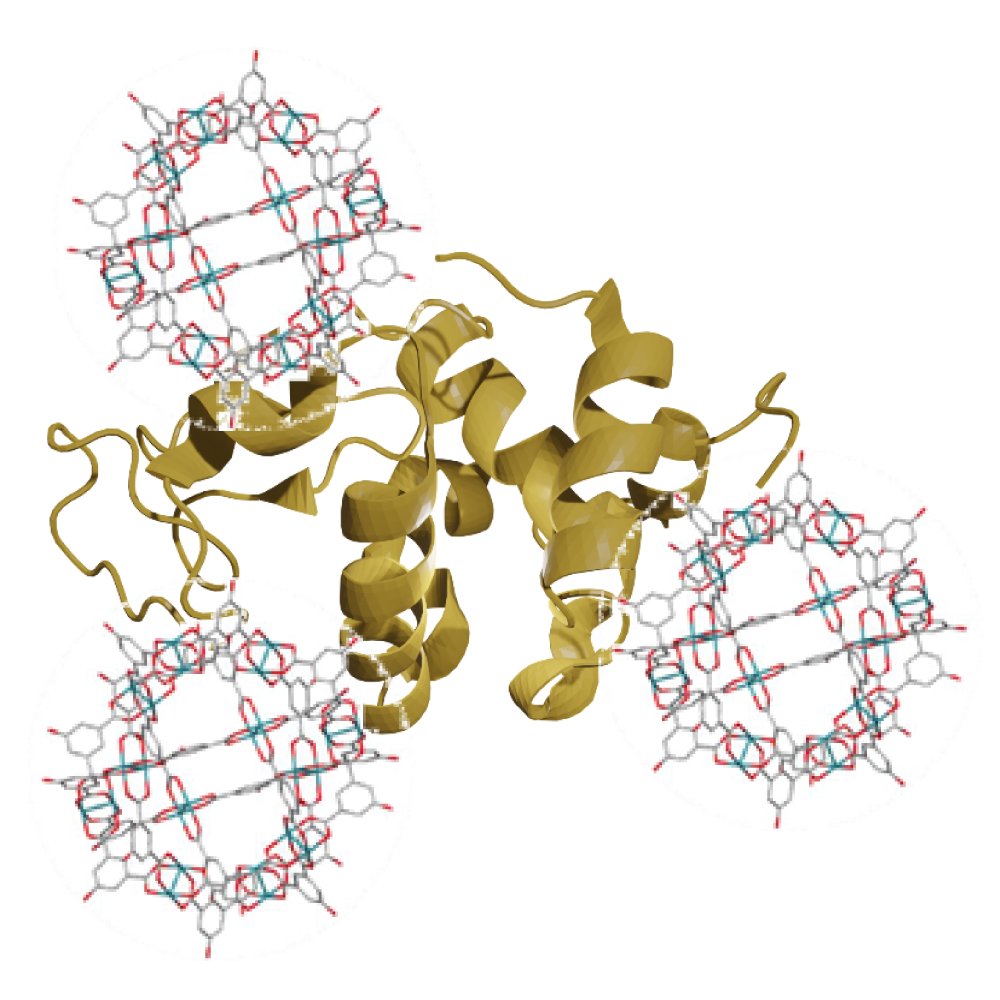
In theme II, we aim to develop novel functional materials by complexing functional metal complexes with polyoxometalate (POM), lipid membranes, and enzymes. For example, we have studied the new functional materials, such as a metal complex with a hollow structure (metal-organic polyhedra (MOP)) complexed with an enzyme to improve enzyme function and recyclability (J. Am. Chem. Soc., 145, 11997 (2023)), the highly sensitive chemical sensor by complexing MOF with an inorganic layered compound, MXene (Chem. Commun., Open Access(2024)), and the construction of highly functional reaction fields by site-specific incorporation of lipophilic metal complexes into liposomes (Chem. Commun., 57, 2895 (2021), etc.).